
Formula Freedom for Families:
Restoring Competition to the Infant Formula Market
Executive Summary
Many parents rely on infant formula to feed their babies. Yet, in the United States, infant formula is expensive, the supply chain is vulnerable, and parents have limited options. The current infant formula market is largely a product of government-driven monopolization through the single buyer rebate program with Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) program. A significant portion of all infant formula purchases are made by WIC, a taxpayer-funded program that aims to provide healthy foods, infant formula, and nutrition education to low-income mothers and children. WIC’s single-buyer rebate program for infant formula awards exclusive state contracts to just one formula company at a time, giving that company a protected market share in the state.
While the intent of the rebate program may have been to lower the cost of providing infant formula through WIC, the result has been quite different. This legal monopoly stifles normal competition, hikes formula prices, creates supply chain vulnerabilities, and leaves parents with few alternatives.
Federal law permits states to implement alternative cost-containment systems for infant formula, yet no state has yet attempted such a program, primarily because of a requirement for economic models projecting cost savings. This paper explains how the problems in the infant formula industry can be addressed and how cost containment can be attained through leveraging market competition and online pilot programs. Future analysis will address the barrier by providing economic models that states can use to meet cost containment.
The “Formula Freedom for Families Act” aims to restore a competitive infant formula market by allowing any willing U.S. Food and Drug Administration (FDA)-approved infant formula manufacturer to provide formula through state WIC programs or allowing WIC participants to obtain a cash-value benefit voucher. Alternative cost-containment approaches improve overall formula costs across the industry, improve supply-chain resilience, and expand consumer choice, including access to healthier options.
Introduction
Infant formula serves as a complete or partial substitute for breast milk, standing as a critical source of nutrition for infants. Though the World Health Organization (WHO) recommends at least six months of exclusive breastfeeding starting from birth, only 24.9% of American mothers are able to meet the standard.1 For long-term health outcomes of both mother and child, exclusive breastfeeding is essential whenever possible.2 But whether due to work constraints, family circumstances, health conditions of mother or infant, or ability to produce breastmilk, three-quarters of all American families depend on access to safe and nutritionally appropriate infant formula as a full or partial substitute.3 Paramount to public health, formula remains a vital resource for families of all circumstances. Yet, the current consolidation of the infant formula market presents significant challenges that impact availability, cost, and consumer choice for all parents.
When government policy drives consolidation, natural competition dwindles. The result is that consumers receive fewer choices, diminished quality, and inflated costs. In the infant formula industry, the same principles hold true, but with heightened risk to babies. As demonstrated by the 2022 formula crisis, if one of the two major formula companies experiences manufacturing troubles, it jeopardizes the health of precious infants throughout the United States. Diversifying formula options will address supply chain issues, lower costs, improve quality, and increase choice. The sections below further expound upon the history of the infant formula industry in the United States, pointing to several sources causing industry consolidation, and expounding upon the risks that arise from these trends.
How the Single-Buyer Rebate Program Creates Market Consolidation
Two formula companies, Abbott and Mead Johnson, dominate 90 percent of the U.S. baby formula market.4 This is because the WIC single-buyer rebate program favors large manufacturers and grants them effective monopolies over state markets. Though large companies are vital for providing many of the specialized types of formula, it also means that smaller manufacturers face difficulties entering the market for some of the more common types of formula. Prior to 2022, the FDA had not registered a new manufacturer in 15 years.5
Through time, more American families have come to rely on infant formula. By the late 1980s, the federal government sought ways to ensure that low-income families could access needed formulas while also keeping program costs down. To that end, in 1989, the federal government enacted a single-payer rebate program for infant formula contracting, which required states to solicit bids from formula manufacturers.6 The manufacturers who win the bid provide the products in exchange for being the only company allowed to sell to WIC participants. State WIC agencies administer the program and monitor access issues.
To be eligible for bidding, the manufacturer must be capable of producing iron-fortified infant formula in powder, liquid, and ready-made forms with both milk-based and soy-based options. Though WIC should secure a variety of formulas for diverse nutritional needs, the requirement means that only large manufacturers can win the contract. By accepting only a single buyer, the requirement consolidates the market by excluding smaller companies that cannot produce all the required forms.
WIC provides formula for nearly 40 percent of infants in the United States and accounts for an estimated 56 percent of all formula consumed.7 This disproportionate consumption by WIC participants, who are more likely to use formula than to breastfeed, amplifies the influence of the single-buyer rebate program on the market. In addition to guaranteed exclusivity, the manufacturer with the rebate contract experiences significant spillover effects from non-WIC participants. A 2025 Government Accountability Office report found that the winner of the single-payer rebate contract accounts for 71–80 percent of a market share.8
Providing all 12 types of formula stands as a significant barrier to entry for smaller formula manufacturers who may only have the capacity to scale to one or two types of formula, according to reports from the Federal Trade Commission and National Academies.9–10 Additionally, state WIC agencies often enter into multi-state bids to ease administrative concerns, which means manufacturers must be capable of supplying multiple states. As such, the single-payer rebate program greatly distorts the market by pushing out new competitors and smaller businesses. Smaller manufacturers often lack the scale of larger incumbent manufacturers, effectively closing them off from the WIC consumer market and any spillover sales.
Manufacturing companies that win the WIC bid secure market dominance, shaping supply chains and geographic concentration. Only three companies, Abbott Nutrition, Mead-Johnson, and Nestle, have the capacity to create all types of formula, in both milk-based and soy-based forms.
All factors combined have led to a significant increase in consolidation of the infant formula market over time.
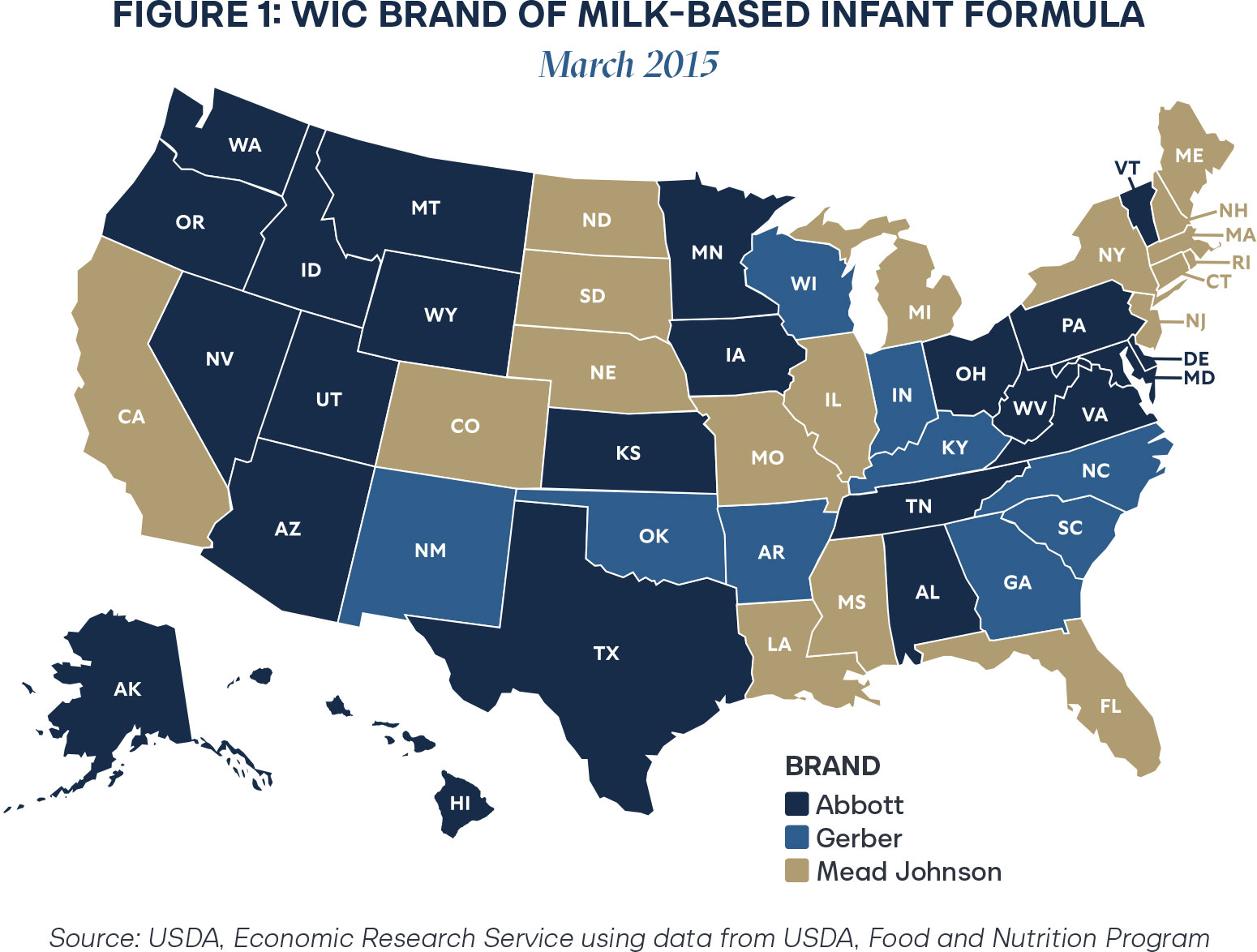
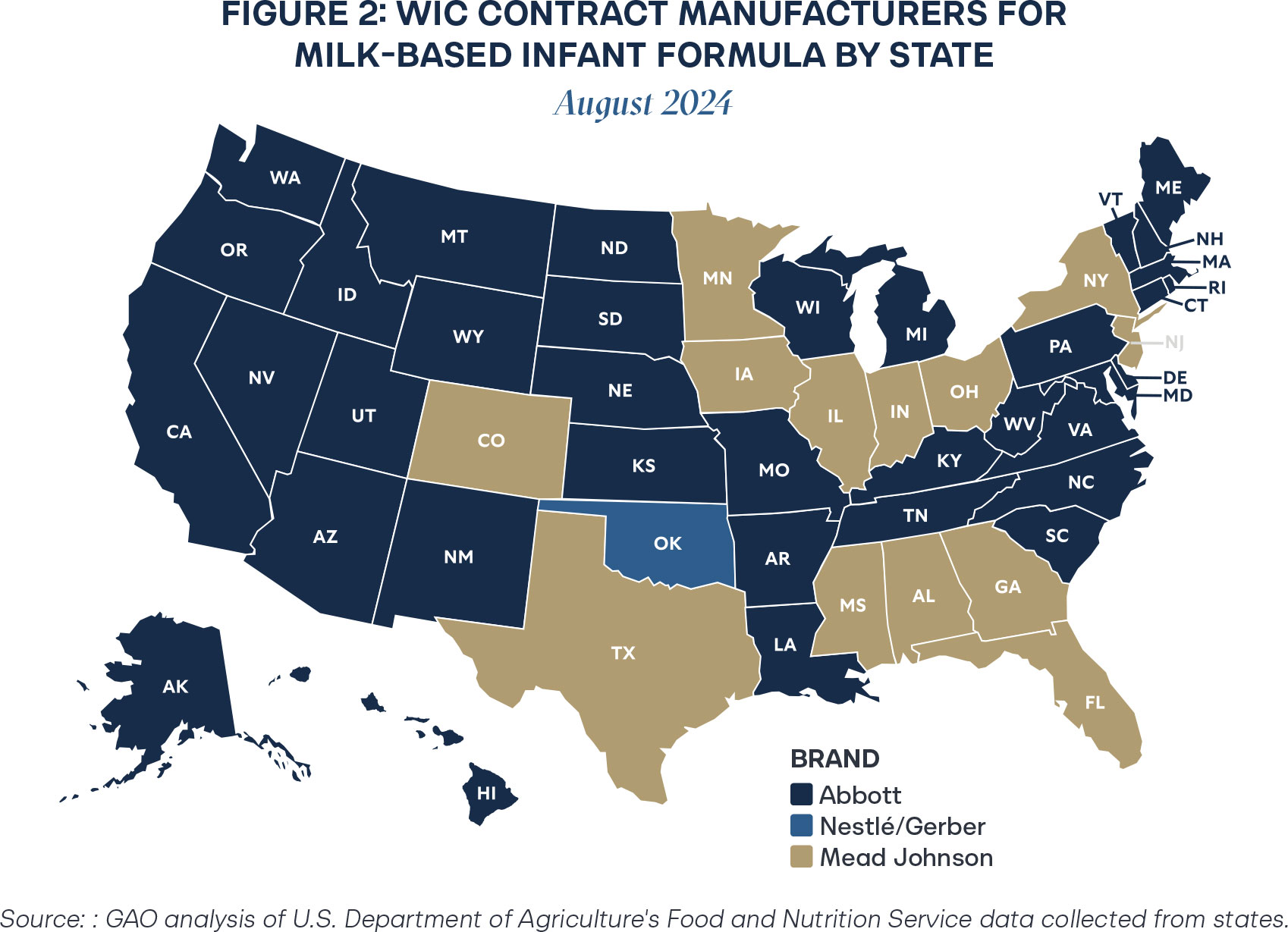
At the end of 2024, Oklahoma transferred the rebate to Abbott Nutrition, further exemplifying the trend of concentration in WIC rebate contracts.11 As of 2025, just two manufacturers provide infant formula to all infants in the United States, which creates fragility in the system in the event of a crisis.
Consolidation Problem 1: Safety and Access Risk
Reliance on just a few manufacturers generates perilous vulnerabilities in a supply chain. In 2022, Abbott Nutrition’s major manufacturing plant in Michigan shut down due to bacterial contamination and insanitary conditions.12 Infant formula around the country was recalled, resulting in the 2022 formula shortage.13 States contracted with Abbott Nutrition for the WIC rebate program were particularly impacted, as winning contracts control around 71–82 percent of a state’s market when accounting for spillover effects.14 Additionally, the regional concentration exemplified in Figures 1 and 2 further complicates supply chain challenges.
Since WIC rebate contracts essentially guarantee a significant portion of the market to one manufacturer, it is not in the interest of losing bidders to establish sufficient supply chains to service a state, as it will not be profitable to do so. That effect is especially compounded by geographic challenges, with Abbott Nutrition rebates clustered largely on the West Coast and Mead-Johnson covering much of the deep South. Though monopolization creates predictability for a single large company, when the unpredictable occurs, companies struggle to respond effectively to public needs. In the case of the infant formula supply, delays threaten little lives.
In a more natural market without rebates from state agencies, many more manufacturers would proliferate. They would compete on the basis of cost, quality, access, and consumer appeal. With truer competition, manufacturers would be incentivized to build the capacity to scale in the event that their formula gains popularity. Without guarantees, manufacturers could plan better supply chains that provide structure for agile response in the event of an emergency.
During the 2022 infant formula crisis, the U.S. market could not scale quickly enough to provide formula to families. The public soon turned to foreign infant formula to supply needs, but regulations from the Food and Drug Administration obstructed access.15 After intense pressure and delays, Congress temporarily suspended tariffs on imported formula and eased strict labeling requirements.16—17 These measures were temporary, with tariffs and labeling challenges now back in place. Permanently easing pathways to allow European formulas that meet FDA nutritional standards in the U.S. market represents another measure to secure consistent access to vital foods.
The crisis neatly exemplifies the current fragility of the infant formula market. Such an emergency could happen again at any moment, whether through contamination at a manufacturing plant, a cyber-attack, national emergencies, natural disasters, or other unforeseen events. Concentration in the formula market only exacerbates shortages when manufacturing challenges occur.
Consolidation Problem 2: Increased Cost
A competitive market is essential to ensure a wide range of affordable options are available to parents, regardless of income. The WIC program plays an important role in securing access to infant formula for low-income families. Having a child is expensive, and for infants exclusively or partially bottle-fed, formula costs can represent the greatest expense of raising a newborn baby. Though WIC participants do not pay out-of-pocket for formula, the current rebate program drives up the cost of infant formula in the overall industry, affecting all families who purchase formula.
When receiving bids for a rebate, states often join cooperative multi-state contracts to ease the administrative burden of procuring infant formula. For example, NASPO Valuepoint, a nonprofit cooperative procurement program that helps states access competitively solicited contracts, procured an agreement with Abbott-Nutrition for 14 states in 2025.18 While perhaps effective for administrative challenges, economic problems linked to the consolidation of rebates can drive up prices across the states.
Rebates operate as a retrospective discount provided by the manufacturer. After a product is purchased, the manufacturer “pays back” a predetermined portion of the purchase price to a middleman. In the case of infant formula, the manufacturers would be Abbott Nutrition or Mead Johnson, and the middleman would be WIC agencies.
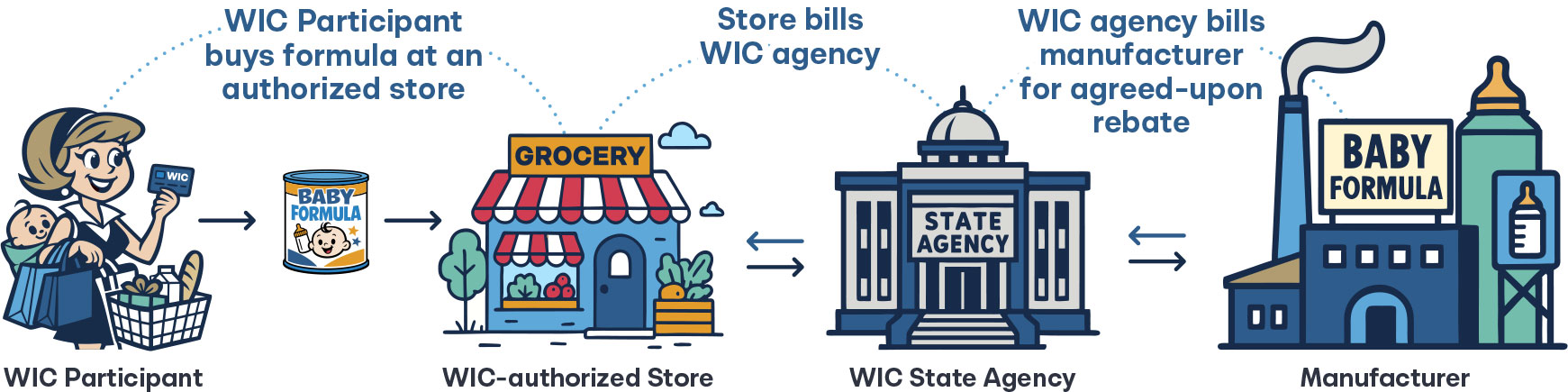
These rebate arrangements are widely criticized as they increase overall costs and encourage perverse incentives. Manufacturers artificially inflate list prices to make rebates seem more generous, which leads to higher prices for consumers and unnecessary costs to the system overall. Nowhere is this felt more keenly than in the pharmaceutical industry.
Drug manufacturers negotiate rebates with pharmaceutical benefit managers (PBMs), who then develop formularies of what the pharmacies in their network can dispense to insured patients. Ideally, these formularies would prioritize cost savings for patients. Rather than favoring lower-priced generics, PBMs often include higher-priced name-brand drugs to maximize rebates from manufacturers. In turn, the manufacturers inflate list prices to sustain larger rebates while maintaining profits. On the whole, the rebate system creates opaque pricing across the system for all parties and misaligns incentives for both PBMs and manufacturers, which often prioritize rebate-driven revenue over patient affordability. Vertical integration within the industry (PBMs owning pharmacies, or insurers owning PBMs) further undermines price transparency. One 2020 study found that a $1 increase in rebates correlates with a $1.17 increase in list price.19 In other words, there is nearly a 1:1 relationship between drug rebates and list prices.
Examining infant formula contract agreements lends insight into a potential estimate of the markups in the infant formulas participating in the WIC program. Simple calculations from the information provided in the publicly available 2025 NASPO agreements reveal percentage markups on formula products offered through the WIC program. For the full agreement with the calculations, see the appendix.
The percent mark-up wholesale was calculated by subtracting the net cost per unit from the wholesale price and dividing by the wholesale price, and the percent mark-up overall was calculated by subtracting the net cost per unit from the retail price and dividing by the retail price.
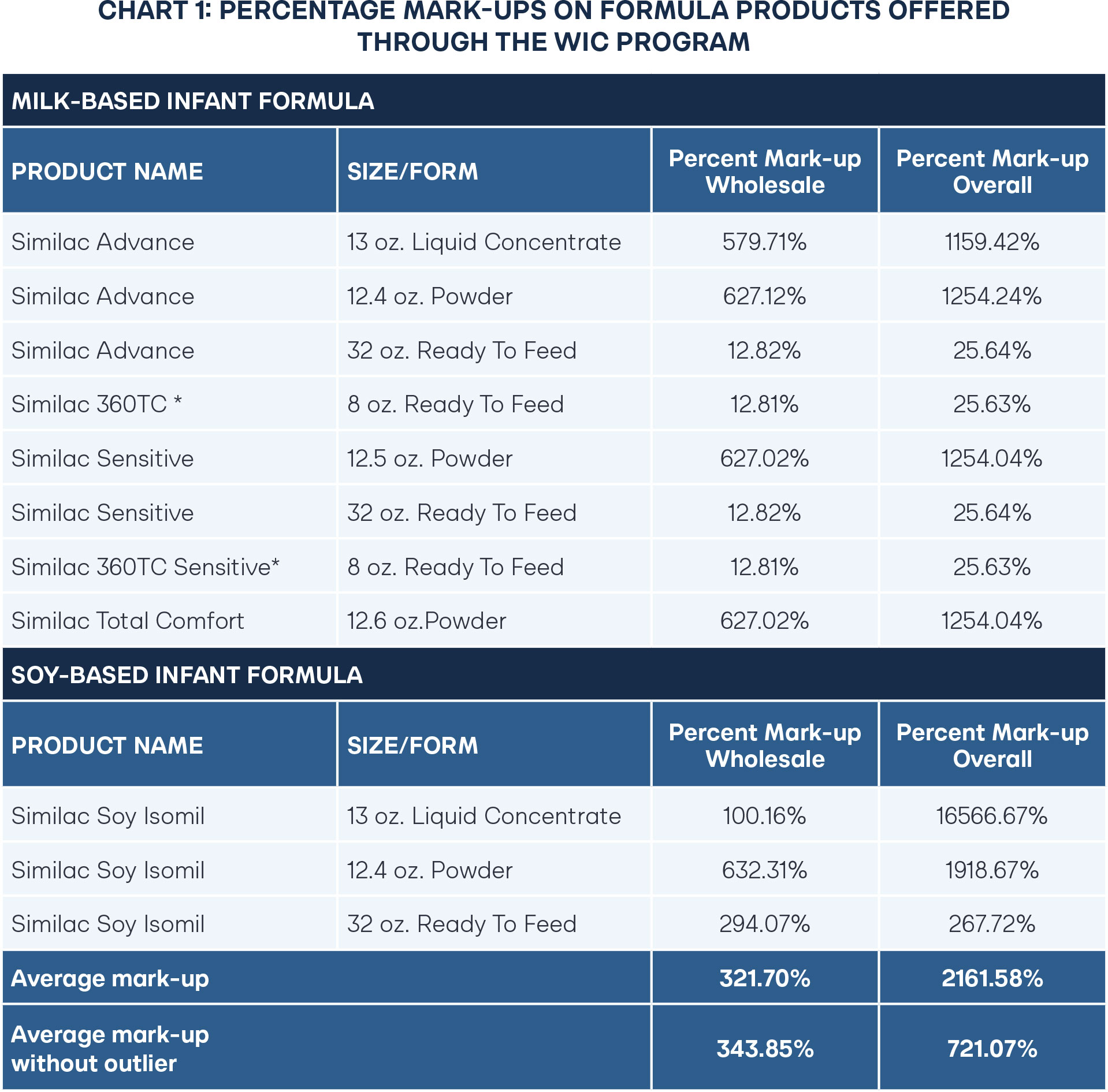
The average overall mark-up for retail prices of infant formula is 721 percent, but if accounting for wholesale prices, then the markup is reduced by about half at 344 percent. One outlier, Similac Soy Isomil liquid concentrate, is uniquely high because it is cheap to produce but can have challenges with storage. Yet two of the most common formulas, powder-based milk and soy formula, are shelf-stable, relatively cheap to produce, and easier to transport, still remain at the high end of wholesale markups at 627–632 percent.
To contextualize infant formula markups with other products, the graph below compares infant formula to common high-priced grocery products and common drugs.

Because of produce spoilage and increased labor costs from deli departments, grocery stores tend to charge more for produce and deli meats. Name-brand snacks also purchase shelf space, and name recognition often means grocery stores can charge more for those products. On the other hand, insulin, inhalers, and many specialty generic drugs necessary to sustain life can be produced at a low price and can be found in generic versions.
Though infant formula is a food product, the markups most closely resemble trends in the pharmaceutical industry. Notably, produce, name-brand snacks, and deli meat are not subject to rebate programs; pharmaceutical products are. The single-payer rebate program for infant formula is likely the culprit driving the high costs of infant formula across the industry.
In the pharmaceutical industry, price reform is often stagnated because pharmacies fault the PBMs, who blame the manufacturers (or vice versa) for high prices. Likewise, the infant formula conversation will likely revolve around WIC pointing at manufacturers for high prices, with manufacturers attributing increases to retailers, and so forth. Regardless, unpacking who is “at fault” can be a fruitless endeavor. Policymakers should understand that rebate schemes represent the heart of the problem. Reforms that inject competition, foster transparency, and improve accountability for rebate programs will do far more to lower prices than targeting specific players.
Consolidation Problem 3: Limited Options—Health and Nutrition Concerns
Consistent with economic principles, when competition deteriorates, so does quality. New infant formula companies rarely appear in the United States because of significant barriers to entry. To increase revenue, manufacturers seek cheaper, lower-quality ingredients with little risk of a new entrant threatening profits by providing formulas with better quality ingredients.
American infant formula often contains corn syrup, added sugars, harmful concentrations of seed oils, and relies primarily on plant fat rather than milk fat.26,27,28,29 Many of these ingredients can harm infant health, but are cheaper to produce and acquire than their healthier counterparts. Silence on FDA standards regarding what qualifies as a suitable fat or protein ingredient means that very few companies in the United States offer formulas without the cheaper and more harmful ingredients. The FDA’s nutrition standards specify minimum and maximum levels for various nutrients, including protein, fat, linoleic acid, vitamins, and minerals. However, what qualifies as an acceptable protein or fat is not explicitly limited by FDA standards, often leading to the use of the cheapest substitutes.30 (See 21 C.F.R. § 107.100)
Prior to 1980, American infant formula had no mandated nutritional standards, and manufacturers generally followed recommendations from groups like the American Academy of Pediatrics.31 In 1980, Congress passed the Infant Formula Act of 1980, which mandated 29 nutrients for infant formula.32 However, a comprehensive review of nutritional requirements for American-manufactured infant formula has not occurred since 1998, despite the fact that the 1980 law requires, “the [HHS] Secretary to…establish requirements for quality factors for infant formula to the extent possible consistent with current scientific knowledge.”33–34 Project Stork Speed, launched by HHS in 2025, finally intends to review the FDA’s nutritional standards for infant formula.35 The agency put out a Request for Information on nutritional standards for infants, but no standards have yet been updated.36
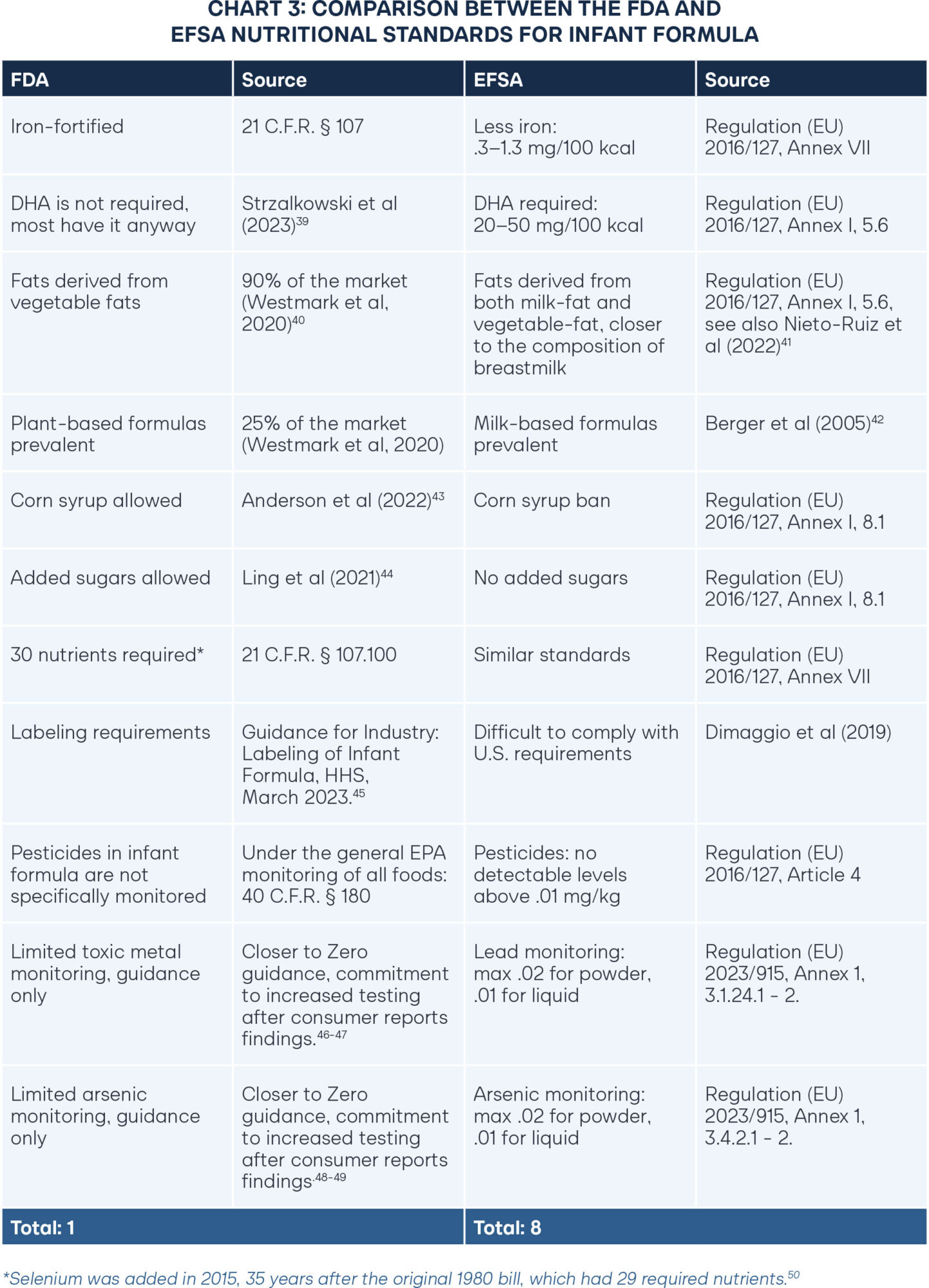
To open the market to healthier options, free-market advocates argue that the FDA should ease pathways for European formulas to sell to Americans. Detractors respond by saying foreign formulas do not meet FDA standards and therefore must not be safe for consumption. The European Food Safety Authority (EFSA), which is Europe’s equivalent of the Food and Drug Administration, reviews the scientific literature on infant formula every few years and updates standards accordingly. EFSA formula standards were last updated in 2016, and before that in 2013.37 Furthermore, a 2019 study reviewed 16 European-manufactured formulas.38 It found that 15 of the 16 formulas met FDA nutritional standards, though none met the FDA’s stringent labeling requirements.
While European nutritional requirements for infant formula have been updated consistently in accordance with current scientific standards, American nutritional requirements have not changed since 1998. A comparison between the FDA and EFSA nutritional standards for infant formula reveals key differences. Better nutritional policies are highlighted in green, with a full tally of beneficial policies for each regulatory agency in the final row.
39 https://smallbusiness.chron.com/supermarkets-determine-markup-produce-cigarettes-80905.html
40 https://doi.org/10.3390/nu12103136
41 https://doi.org/10.3389/fnut.2022.820224
42 https://doi.org/10.1097/00005176-200002000-00006
43 https://doi.org/10.1093/ajcn/nqac173
44 https://doi.org/10.1093/jn/nxab044
45 https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-infant-formula-enforcement-discretion-policy
46 https://www.fda.gov/food/environmental-contaminants-food/closer-zero-reducing-childhood-exposure-contaminants-foods
47 https://www.consumerreports.org/babies-kids/baby-formula/baby-formula-contaminants-test-results-a7140095293/
48 https://www.fda.gov/food/environmental-contaminants-food/closer-zero-reducing-childhood-exposure-contaminants-foods
49 https://www.consumerreports.org/babies-kids/baby-formula/baby-formula-contaminants-test-results-a7140095293/
50 https://www.federalregister.gov/documents/2015/10/13/2015-25960/infant-formula-the-addition-of-minimum-and-maximum-levels-of-selenium-to-infant-formula-and-related
Several differences between U.S. and European formulas raise concerns for public health. Allowing corn syrup and added sugars in infant formula is associated with significantly higher rates of childhood obesity. A 2022 study focused on infants in the WIC program found that of the infants who received formula made with corn syrup, 25 percent were obese by age four.51 This outcome alone represents a deeply troubling public health concern. Another study found that added sugars lead to rapid weight gain, as early as nine months, and added sugars for formula-fed infants double that of sugar consumption of breast-fed infants.52

Regular testing for poisonous substances such as lead and arsenic should already be a given, considering the inherent vulnerability of small infants. Yet in 2025, Consumer Reports discovered high levels of lead and arsenic throughout the formula supply. As David Carpenter, the Director of the Institute for Health and the Environment at the State University of New York at Albany, said, “There is no excuse for having arsenic in baby food or formula.53 Absolutely none. The industry should be bending over backward to be certain it’s not there.” The discovery prompted HHS Secretary Kennedy to announce “Project Stork Speed,” with an increased commitment to regular testing on the infant formula supply.
Infants are especially susceptible to even small quantities of pesticides.54 In the United States, the EPA regulates pesticide levels in infant formula using the same guidelines for all foods. Given the relative size and sensitivity of infants, there should be stricter limits and closer monitoring of pesticide residues in infant formula.
For the fat composition of formula, a complex blend of milk fat and vegetable fat most closely represents breastmilk.55 However, the majority of U.S. formulas rely on vegetable oils entirely for the fat content since vegetable oils tend to be cheaper. Though many U.S. formulas include milk-based ingredients, the milk often comes from skim milk and serves as the protein. Relying on fat from whole milk is particularly important because it contains Milk Fat Globule Membranes (MFGM), which is also present in breastmilk and crucial for brain development.56 Brands such as Bobbie and Kendamil have recently emerged in the American market to provide whole-milk options, but WIC participants do not have access to them, furthering socioeconomic divides in health outcomes.57–58
While updating nutritional standards at the federal level is a necessary step toward improving the quality of infant formula in the United States, it alone is not a complete solution. Injecting competition into the infant formula market, both through domestic reforms and easing pathways for foreign manufacturers, would incentivize companies to not only meet but exceed FDA quality standards. In a competitive market, quality and innovation drive success.
The Solution: Formula Freedom for Families Act
Reintroducing competition in the infant formula market would strengthen the supply chain, decrease costs, increase options, and improve quality. Some may assume that expanded competition would only be possible by removing the single-buyer rebate program for WIC, which distorts the market, but is currently required by federal law. However, changing federal law to remove the program would be a formidable lift politically due to cost and access concerns.
This political challenge suggests that alternative state solutions are necessary. States can implement reforms that do not require federal legislative changes and can serve as a testing ground to demonstrate that increased competition in the industry would improve infant formula for everyone.
Though federal law requires a single-buyer rebate program, it also allows states to adopt an alternative cost-containment system under 7 C.F.R. § 246.16a(d). No state has yet attempted such a program, partly because states must project food cost savings under an alternative cost-containment system.
The “Formula Freedom for Families Act” aims to restore a competitive infant formula market by providing states with two options for alternative cost containment programs. The purpose of the act allows states to maintain competitive rebate pricing while simultaneously restoring competition to the market, which will drive down the cost of formula for all parents, improve supply-chain resilience, and expand consumer choice. Addressing issues in the single-payer rebate program will also help WIC to save more money as well, a true win for both participants and state governments. The section below outlines each alternative cost containment program and specific strategies states can take in implementation.
Alternative Cost Containment Program Option 1: Accept Any Willing Provider
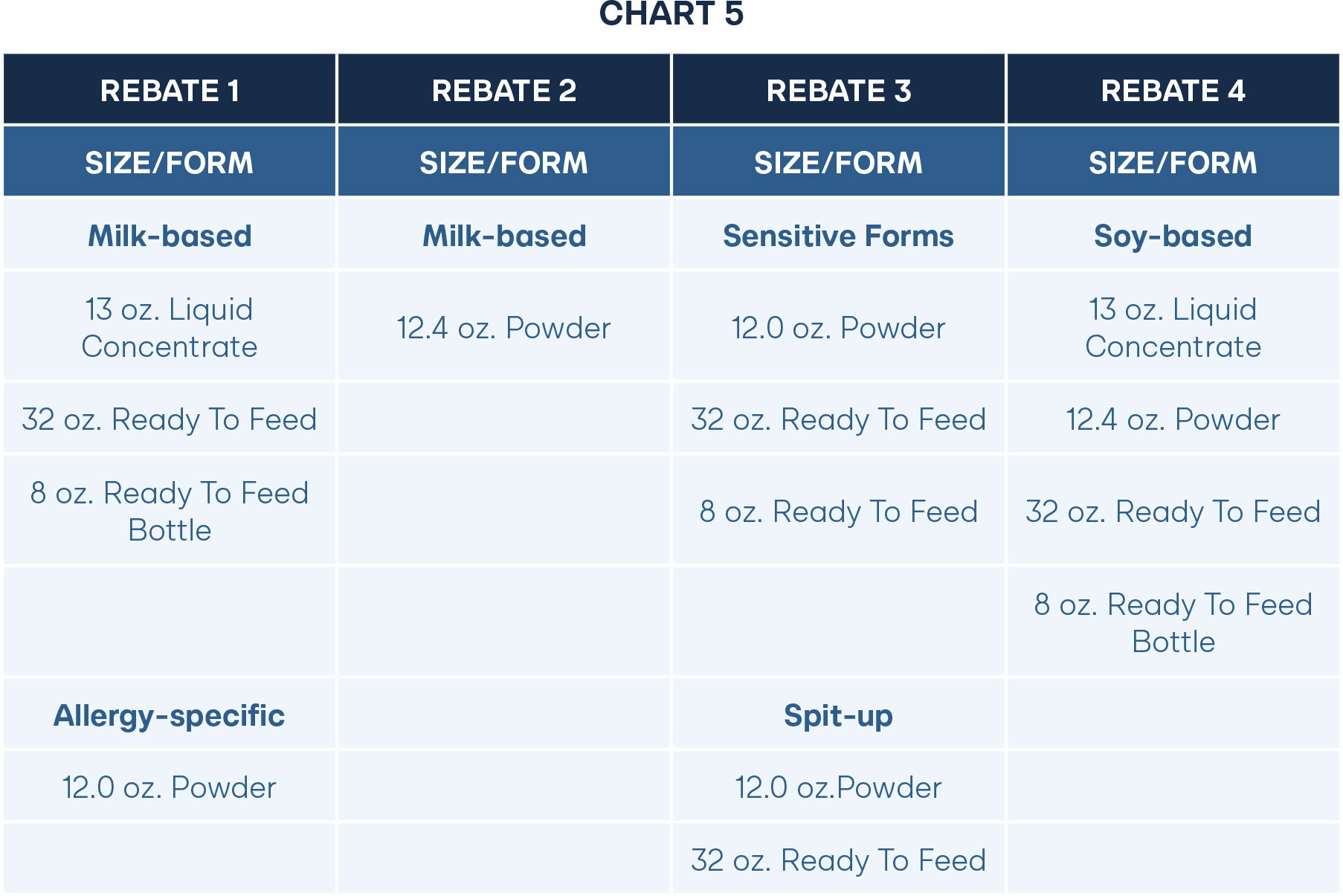
Under the current structure of the single-buyer rebate program, states only accept one manufacturer for all types of formula in both milk-based and soy-based forms. This prevents smaller manufacturers that only have the capacity to produce a few types of formula from winning a rebate. State WIC agencies should remove any requirement that infant formula manufacturers must produce and provide all available formula forms currently required by the state WIC agency (including but not limited to powder, liquid concentrate, and ready-to-feed formulas in both milk-based and soy-free forms) as a condition of rebate participation.
Instead, states should allow any willing provider that meets legal nutritional and safety standards to win a contract on a specific formula type. For example, states could accept a competitive rebate from a smaller company for the milk-based powder formula. But for the other types, such as liquid concentrate and ready-to-feed forms, the typical single-buyer manufacturer would offer rebates over the majority of products. This means the state could still secure a generous rebate while also allowing a smaller manufacturer to provide formula to WIC participants. Since a smaller manufacturer would be motivated to gain customer recognition for a new group, they will also likely desire to offer a lower rebate than the rebate for the existing company. Additionally, a smaller company is more likely to have developed infant formula with better ingredients, which could appeal to WIC agencies and help motivate the larger companies to improve quality to secure contracts.

Alternatively, a state agency could strategically accept rebates from several manufacturers over a wide variety of products. Rebate amounts differ significantly depending on the product. For example, in the 2025 NASPO Infant Formula Rebate Agreement with Abbott Nutrition, the discount for milk-based powder was 86 percent. On the other hand, the percent discount for ready-to-feed milk forms was only 11 percent. By opening the possibility for multiple contracts, large and small manufacturers would compete on who could provide the largest discount on products, and not on who can produce all types. Accepting offers for discounts on each product would drive down the overall cost for WIC and in the general market.
Notably, this version would also introduce administrative complexity on the front end for WIC agencies, which could result in confusion for participants. Participant confusion could be easily addressed by developing an online pilot program where parents take a quiz that identifies which formula they need and delivers it to them. Or, instead of an online program, WIC agencies could house infant formula at local agencies rather than at traditional retailers.
Expanding the pool of contracted manufacturers will provide WIC participants with increased options for infant formula. This can help drive down costs for the program and improve quality through greater access to formulas with different nutritional profiles, such as those without corn syrup or an over-reliance on vegetable oils. Enabling new entrants will restore competition and diversify infant formula options for all.
Alternative Cost Containment Program Option 2: Cash Value Benefit Vouchers
As an alternative to the single-buyer rebate contracts, the state WIC agency could establish a cash value benefit voucher system. This system would allow participants to purchase any FDA-approved infant formula products available at WIC-authorized retailers.
The full value of the voucher would be applied toward any infant formula product for which the state has an active rebate agreement. If a recipient wishes to use the voucher on an FDA-approved formula without an active rebate agreement, the voucher would cover an amount equal to the state’s rebate for that formula type, with the recipient responsible for any remaining balance.

The voucher may be used for any FDA-approved formula product and would be exempt from sales taxes. Rebate amounts and covered formulas would be clearly displayed on the voucher. Additionally, the voucher could be combined with coupons provided by the retailer or manufacturer. For example, with the adoption of a cash value benefit program, smaller manufacturers would be motivated to provide coupons to WIC participants to gain a larger consumer base. Alternatively, retailers may want to sell certain non-WIC products faster and could offer markdowns that could be combined with the cash value benefit voucher.

Since WIC participants rely on government assistance for food benefits, it is likely that many will not want to pay anything out of pocket. For this reason, many will continue to use WIC brands. But creating this pathway allows all parents, regardless of financial circumstance, a greater ability to access any type of formula. It creates freedom and encourages smaller manufacturers to compete for a new consumer base.
To reduce potential participant confusion, WIC agencies should create educational materials and clear vouchers that participants can use.

Properly implementing the cash value benefit voucher program could be challenging for WIC-approved vendors. However, it has been done before by the Supplemental Nutritional Assistance Program (SNAP) with cash value voucher programs for fruits and vegetables. Over half of all states have adopted a “Double Up Food Bucks” program, which allows SNAP recipients to purchase double the amount of fruits and vegetables.59 For example, if a participant has $5 to spend in SNAP, they can get $10 worth of products if it is spent on fresh produce. Due to the extra complication, participation is optional for vendors. By giving vendors the option to participate, some may not view training as worth the effort, while others will want to ensure implementation through added sales.
Benefits of an Online Model
Alternatively, vendor compliance can be improved, and the potential of participant confusion can be reduced by offering cash value benefit vouchers through an online program. Instead of purchasing formula at a traditional retail store, participants could go to an online portal and answer a questionnaire to determine which type of formula they would like to purchase. Once the type and form were identified, participants would then be given the option to receive the brand with the rebate at no cost or pay a little extra for an alternative brand. Large companies with robust online retail structures, such as Amazon, Walmart, or Kroger, could help facilitate the program.
Since WIC manufacturers must provide infant formula in all WIC-approved vendor locations, they must create an oversupply. This further drives costs and may account for a portion of the high markups. Adopting an “warehouse model” where a large vendor houses formula in a centralized location and then mails it directly to participants would improve access, improve vendor compliance, and decrease overall program costs.
Conclusion
Reforming WIC’s contracting process through the adoption of the “Formula Freedom for Families Act” will mitigate future infant formula shortages, improve infant formula quality outcomes, and decrease costs by enhancing market competition. By allowing any willing provider to participate in the rebate program or allowing cash value benefit vouchers, states can foster a more resilient, healthy, and consumer-friendly infant formula market.
Appendix
The dataset linked below reflects the percent markups for each product included in the
2019 and 2025 NASPO rebate agreements.
Acknowledgments
Thank you to Adam Meier for his encouragement and continual advice throughout the development of this paper. To Ryan Quandt, thank you for his review of potential cost-containment strategies and his ongoing efforts on this issue. Thank you to Josie Pearce for the review on the nutritional quality of infant formula. Finally, thank you to my sweet daughters who motivate me every day to help improve infant formula for more children like them.

Stay Informed
Sign up to receive updates about our fight for policies at the state level that restore liberty through transparency and accountability in American governance.
